|
Pre-laboratory Assignment: Introducing Measurements in the Laboratory T he unknown sample and the capillary tube (together) should be disposed of in the labeled waste container provided when you are finished.Also note that melting points depend on the purity of the solid. The melting points given in the table represent the lowest temperature in that range, where liquid formation is first observed. Please note that organic solids actually melt over a range of temperatures. * This melting point data was obtained from the NIST Standard Reference Database Number 69 ( ). Then evaluate the accuracy in your measurement by calculating your percent error. Identify your solid by comparing your experimental melting point with the true melting points supplied. Your unknown solid is one of the substances listed in the table below.You will also need to obtain and record the melting points that they have measured. Share your measured value with all other groups who were assigned the same unknown solid as you.The melting point is the temperature at which liquid first appears.Stir the bath continuously and watch your sample carefully. The flame should be adjusted to a moderate temperature, with its tip touching the bottom of the beaker. Heat the water bath slowly with your Bunsen burner. 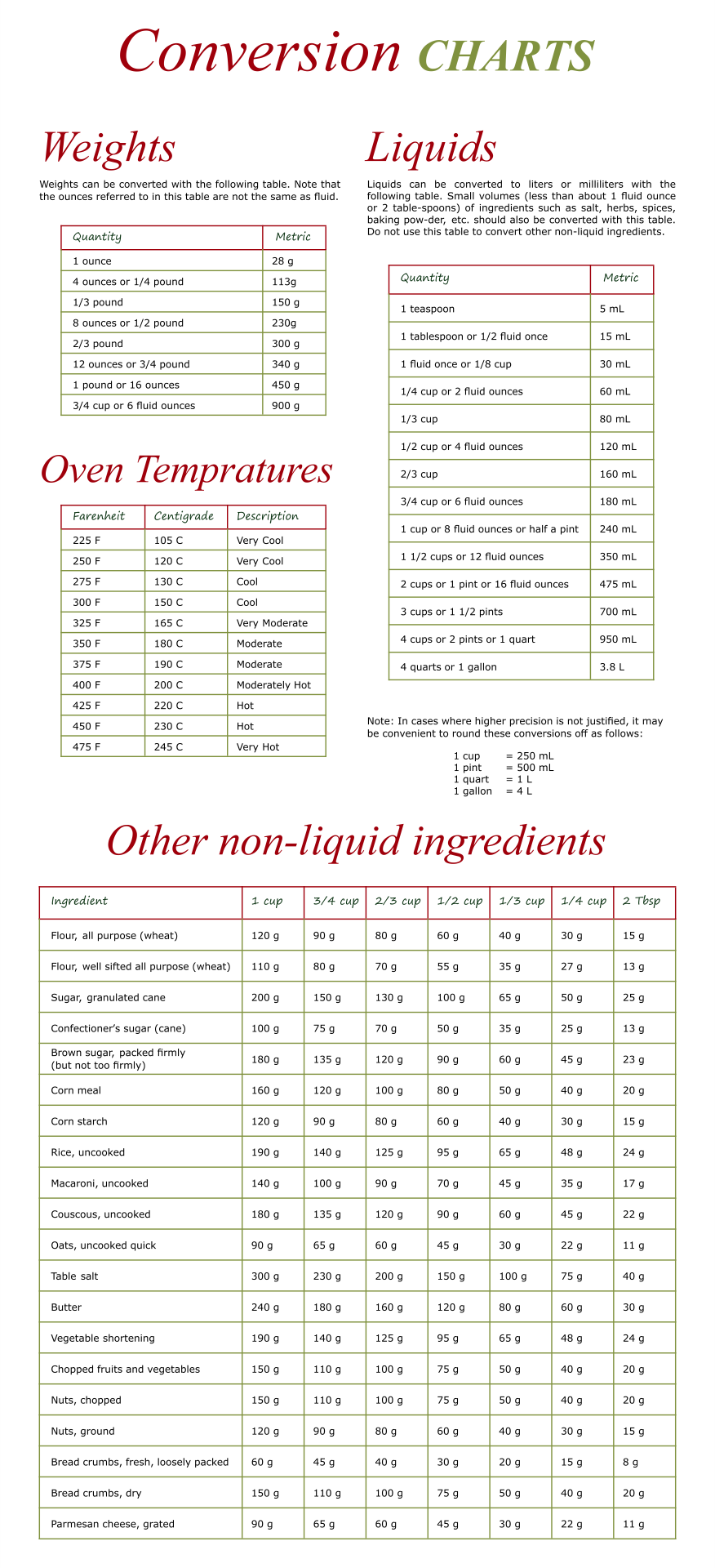
Place a slotted stopper around the thermometer, and using a clamp, suspend it in the water bath.The sealed end should be close to the bulb of the thermometer. Use a small piece of latex tubing like a rubber band to attach the capillary tube to your thermometer.Use a large 400-mL beaker half-filled with tap water for the hot water bath.Assemble your equipment as shown in the diagram on page 5.Repeat until you have a depth of about 2-mm of solid in the tube. Then turn the tube over and tap the tube lightly against the lab bench to allow the powder to fall into the sealed end. Press the open end of the capillary tube into the powder. Obtain a capillary tube from your instructor.Powdered samples of the unknown solids are located at the front of lab on the instructor’s lab bench. Record the ID code of the unknown solid assigned to you and your partner.Part D: Measuring the Melting Point of an Unknown Solid Explain it to them by using these charts and tables.\( \newcommand \times 100\]Īccurate measurements will typically have low percent errors of <5%. So, make this easier and simpler for your children too. It is also introduced and taught in third and fourth grade. The spoons used for daily measurements are ⅛ teaspoons, ¼ teaspoons, ½ teaspoons, 1 teaspoon, and 1 tablespoon.Īpart from cooking, the conversion chart is used in agricultural experiments and other scientific experiments. The calibrated measuring cups made up of ¼ cup, ⅓ cup, ½ cup, and 1 cup sizes are available. Steel spoons are used wherever the spoons are mentioned in the said chart. According to the liquid measurements chart, the liquid is to be measured with a clear glass or plastic cup that has a pouring spout. These charts can help you a lot in your daily cooking sessions to make accurate measurements. For instance, 4.93 mL is rounded off to 5 mL, and so on. Please Note: The figures mentioned in the charts above contain the approximate values.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
